Daily Directions-->Go to the Google classroom & open the assignment, " Atomic Structure( Revised Student copy) -shergill"
|
No Warmups !
Daily Directions-->Go to the Google Classroom & Work on the Assignment, " Average Atomic Mass Introduction" Daily Directions--> Go to the Google Classroom & work on the Assignment, " Average Atomic Mass Introduction"
1. Describe the changes(s) to a Neutral atom if a proton is added.
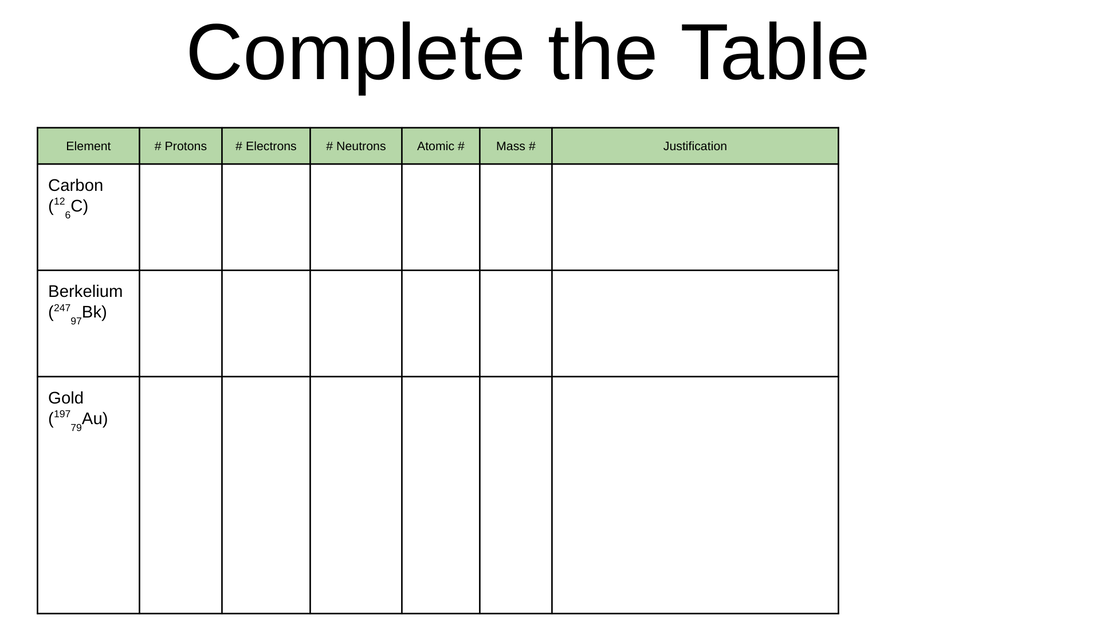
2. Describe the Changes to a Neutral atom if an electron is added. Daily Direction(s)--> Work on any missing or Incomplete assignment/Assessment for this class. Note: Mass number = # protons + # neutrons
In a neutral atom: # protons = # electrons Question: (Look at the periodic table) What element has an atomic number of 24? How many protons, electrons, and neutrons does this element contain? Justify your answer. Daily Directions--> Go to the Google classroom & work on the activity, " Building an Atom Activity" Daily Directions---> Go to the Google Classroom & work on "Penny Lab Activity" (If you pass the Lab safety test with 80% or higher or take the Lab safety test)Daily Directions--> 1. Turn in the lab safety contract and check you took the Quiz on lab safety( Must turn in the contract and pass the safety quiz with 80% or better)
2. Go to the Google Classroom & watch the video on , "Edpuzzle - Atomic #, Mass #, and Net Charge of Atoms 20-21"(Must submit before the class ends to get full points) Daily Directions--> 1. Take the " Lab Safety Quiz" if did not take yesterday.
2. Watch the video on , "Edpuzzle - The Atomic Structure" ( Must take detailed notes and answer all the questions embedded within the video to get points.) |
DirectionsAnswer the questions in the corresponding sections of your warm up sheet
|